
Department of Insect Symbiosis
Director: Prof. Dr. Martin Kaltenpoth
The Department of Insect Symbiosis investigates how insects adapt to challenging environments or food sources, with a particular focus on ecological traits conferred by microbial symbionts. The main goal is to understand the evolutionary origins, ecological implications, and the chemical and molecular basis of insect interactions with mutualistic microbes, as well as with antagonists and the insects’ food plants. In order to derive general principles governing these interspecific interactions, we study a broad spectrum of insect taxa including diverse species of beetles, bugs, wasps, and flies.
Insects are the most diverse animal class on the planet, having successfully adapted to a multitude of environmental conditions and food sources. Part of their evolutionary success is due the ability to engage in mutualistic interactions with microorganisms that confer novel ecological traits to their hosts by providing nutritional supplementation, digestive or detoxifying capabilities, or defense against natural enemies.
The department’s main goal is to understand the evolutionary origins, ecological implications, and the chemical and molecular basis of insect interactions with mutualistic microbes, as well as with antagonists and food plants. Our philosophy is to approach these questions in a range of non-model organisms with interesting natural histories, aiming at an understanding of the chemical mediators of interspecific interactions and their functional implications from a cellular level to the fitness consequences for the interacting organisms under ecologically relevant conditions. To this end, we use a combination of ecological experiments, phylogenetic reconstructions, state-of-the art fluorescence microscopy, and molecular, chemical-analytical, and microbiological techniques.
Symbiont-assisted cuticle biosynthesis as a key innovation contributing to the evolutionary success of beetles (ERC Consolidator Grant "SYMBeetle")
To elucidate the key adaptations underlying evolutionary success remains one of the central challenges in evolution and ecology. However, rigorous experimental tests are often hampered by the lack of replicate evolutionary events or the inability to manipulate a candidate trait of importance. SYMBeetle exploits the naturally replicated evolution of an experimentally tractable, symbiont-assisted key adaptation in beetles to understand its impact on niche expansion and diversification. Recent evidence indicates that beetles across multiple different families associate with microbial symbionts that provision their host with tyrosine, an aromatic amino acid necessary for cuticle biosynthesis, hardening, and tanning. SYMBeetle addresses the hypothesis that the acquisition of tyrosine-supplementing microbes constituted a key innovation across phylogenetically distinct beetles that allowed them to expand into novel ecological niches, by relaxing the dependence on nitrogen-rich diets for successful formation of the rigid exoskeleton and protective front wings. Specifically, tyrosine supplementation may facilitate the transition to herbivory and allow for subsisting at very low ambient humidity, by improving the production of a thick cuticular barrier to desiccation. To test this, SYMBeetle combines experimental manipulation of symbiotic associations to assess the symbionts’ contribution to cuticle biosynthesis and its fitness consequences (desiccation resistance and defense) with large-scale comparative approaches aimed at elucidating the taxonomic distribution, ecological contexts, and evolutionary origins of cuticle-supplementing symbioses. The results are expected to transform our understanding of microbes as important facilitators for the evolution of herbivory and the colonization of dry habitats in beetles, two factors of major relevance for the emergence of economically relevant insect pests of agricultural crops and stored products.
Participating researchers: Vojtech Beneš; Soumi Bhattacharyya; Ana Simao Pinto de Carvalho; Dongik Chang; Tobias Engl; Roy Kirsch; Ronja Kruesemer; Sreyashi Mandal; Christin Manthey; Camila Fiori Pereira; Heiko Vogel; Juergen Wierz
Collaborators: Dirk Ahrens (Zoological Research Museum Alexander Koenig, Bonn, Germany); Abdelaziz Heddi and Anna Zaidman-Remy (INSA Lyon, France); Jean Keller (CNRS, Université Toulouse III); Aurelien Vigneron (Claude Bernard Université Lyon I); Takema Fukatsu (AIST Tsukuba, Japan)

The impact of symbiosis and horizontal gene transfer on the evolution of herbivory in leaf beetles
Leaf beetles (Coleoptera, Chrysomelidae) are one of the most speciose families of organisms on earth, with more than 40,000 described species. Their evolutionary success is based on the ability to complete their entire life cycle on a plant-based diet. To achieve this, they efficiently break down the plant cell walls by a set of plant cell wall degrading enzymes (PCWDEs), some of which are encoded by microbial symbionts while others have been integrated into the beetles’ genomes following horizontal acquisition from microbial donors. To understand the relative impact of symbiosis and horizontal gene transfer on digestion of plant material during the evolution of leaf beetles, we are aiming at characterizing symbiont- and host-encoded PCWDEs across all subfamilies of Chrysomelidae.
Participating researchers: Ana Simao Pinto de Carvalho; Roy Kirsch;
Collaborators: Adam Slipinski (CSIRO Australian National Insect Collection, Canberra, Australia), Yu Okamura (University of Tokyo)
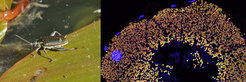
Ecology and molecular regulation of the defensive symbiosis in darkling beetles
Darkling beetles of the subfamily Lagriinae are polyphagous insects with a wide distribution in the tropics and temperate regions. Early morphological descriptions highlighted the presence of specialized organs associated to the reproductive system of females as well as in the dorsal region of larvae, which were believed to harbor symbiotic microorganisms. We identified these bacteria as multiple different strains of Burkholderia gladioli (Beta--Proteobacteria) and confirmed their vertical transmission route from the female reproductive tract via the egg surface into the larval organs, as well as horizontal transmission via the food plant. Manipulative experiments revealed a protective role of the bacteria on the egg surface and in the larvae, highlighting the potential of symbiotic microbes to protect immature life stages of their hosts against antagonists. Since at least some of the Lagria symbionts are culturable and genetically tractable, this system presents an excellent opportunity for elucidating the molecular and chemical basis of host-symbiont interactions as well as their evolutionary origin through a combination of genetic manipulation, bioassays, chemical analytics, genomics, transcriptomics, and phylogenetics.
Participating researchers: Johann de Beer Alina Nick; Heiko Vogel
Collaborators: Laura Victoria Flórez (University of Copenhagen, Denmark); Christian Hertweck (Leibniz Institute for Natural Product Research and Infection Biology, Jena, Germany); Jason Kwan (University of Wisconsin-Madison, USA); André Rodrigues (UNESP Campus de Rio Claro, Brazil)

Symbionts for pathogen defense in beewolves
Beewolves of the genera Philanthus, Trachypus, and Philanthinus (Hymenoptera, Crabronidae) are solitary digger wasps that engage in a symbiotic association with high-GC gram-positive Actinobacteria (‘Candidatus Streptomyces philanthi’). The bacteria are cultivated in specialized antennal gland reservoirs of female wasps and secreted into the subterranean brood cells. The wasp larva later actively transfers the symbionts to its cocoon, where the streptomycetes provide protection against a broad range of opportunistic fungi and bacteria by producing up to 49 different antibiotics. We detected Streptomyces philanthi in about 40 different beewolf species, with the symbionts forming a well-defined monophyletic clade within the genus Streptomyces, suggesting a highly specific association between the wasps and S. philanthi that probably originated in the late Cretaceous (at least 68 million years ago). We are interested in the chemical and molecular interactions between the symbiotic partners of the beewolf-Streptomyces symbiosis as well as the genomic consequences of the symbiotic lifestyle for the bacteria. Additionally, we study how the symbiont-provided defense interacts with the wasp’s own immune defenses, and how the symbionts are protected from the host’s defenses.
Participating researchers: Chantal Ingham (JGU); Bernal Antonio Matarrita Carranza
Collaborators: Jens Rolff (FU Berlin, Germany)

Impact of the microbiome on the viable temperature range of Drosophila melanogaster
Microbial symbionts represent major sources of evolutionary innovation by providing novel ecological traits to their hosts. While the impact of bacterial mutualists on the interaction of insects with their biotic environment (antagonists, diet, etc.) has received increasing attention over the past decades, the importance of microbial symbionts for the adaptation to abiotic conditions remains poorly understood. Preliminary data indicate that microbes associated with Drosophila melanogaster can improve the fly’s cold tolerance in a diet-dependent manner. We aim to understand (i) which microbial taxa that are naturally associated with D. melanogaster enhance cold tolerance, (ii) how they do so on the mechanistic level, and (iii) whether there are seasonal changes in the microbiota of field-collected D. melanogaster that suggest an involvement of microbes in temperature acclimation under natural conditions. We address these questions by a combination of field sampling, cultivation-dependent and –independent microbiota characterization, bioassays assessing cold tolerance, and chemical analytics. We aim to derive insights into the impact of microbial symbionts on the adaptation of fruit flies to low temperatures and their possible role for the success of D. melanogaster in colonizing the temperate regions.
Participating researchers: Marion Lemoine
Collaborators: Marko Brankatschk (TU Dresden, Germany)
The role of bacterial symbionts for bark beetle ecology
Conifer forests in central Europe are currently suffering from an enormous outbreak of bark beetles. Millions of hectares of spruce trees (Picea abies) in Germany and surrounding countries are being killed by the Eurasian spruce bark beetle (Ips typographus) due to warming temperatures that speed up the beetle life cycle and increase tree stress. Successful attack of the spruce bark beetle is known to depend on the presence of free-living fungal symbionts, which the beetle inoculates into the attacked tree. However, bacterial symbionts in the gut could also play an important role in promoting beetle attack by supplying critical nutrients or detoxifying host tree defenses. We are investigating the role of the gut microbial community in promoting bark beetle development on spruce by a combination of culture-dependent and –independent microbiota profiling, fluorescence in-situ hybridization, metagenomics and metatranscriptomics of beetle guts, manipulative bioassays, and chemical analytics. Knowledge about the role of microbial gut symbionts in the bark beetle life cycle may yield new insights into how insects are able to survive on trees with low nutrient content and abundant chemical defenses, as well as suggesting new strategies to combat this pest.
Participating researchers: Ana Patricia Banos Quintana
Collaborators: Jonathan Gershenzon, Department of Biochemistry



